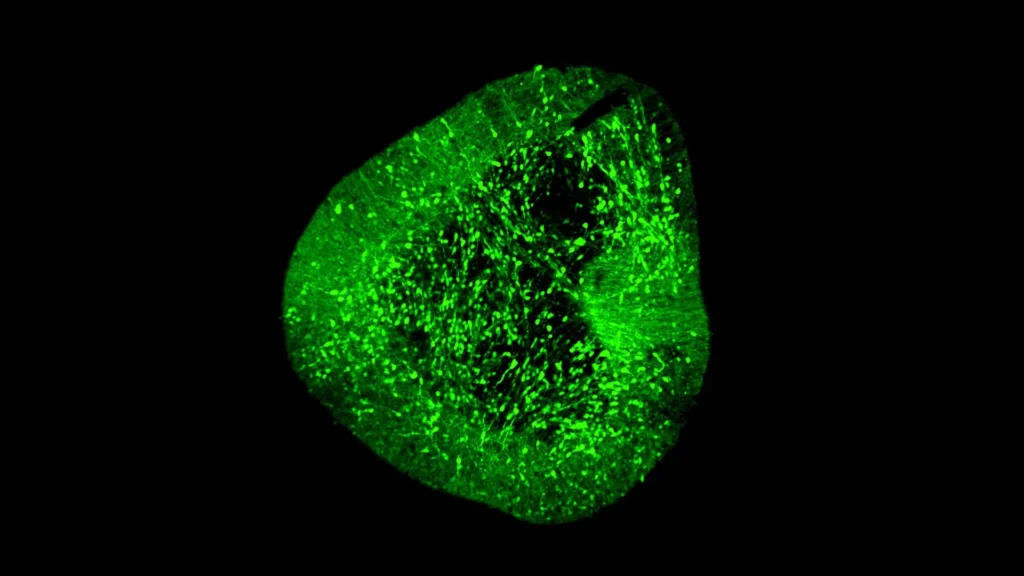
Scientists at Duke-NUS Medical School, in a pivotal collaboration with partner institutions, have successfully assembled one of the most comprehensive single-cell maps of the developing human brain. This monumental achievement, detailed in a study published in Science Advances, represents a significant leap forward in understanding the intricate architecture and developmental processes of the human brain at an unprecedented resolution. The atlas meticulously identifies nearly every cell type present during development, records their unique genetic signatures, and elucidates how these diverse cells grow, mature, and interact within the complex neural environment. Crucially, the research also provides a rigorous comparison of leading laboratory methods currently employed to produce high-quality neurons, directly advancing global efforts to develop novel and more effective cell therapies for debilitating neurodegenerative conditions such as Parkinson’s disease and other severe brain disorders.
Understanding Parkinson’s Disease: A Growing Global Challenge
Parkinson’s disease (PD) stands as the second most common neurodegenerative condition in Singapore, affecting approximately three in every 1,000 individuals aged 50 and above. Globally, the prevalence is even more pronounced, with estimates suggesting over 10 million people worldwide are living with the disease, a number projected to rise significantly with an aging global population. This progressive disorder is characterized by the gradual degeneration of midbrain dopaminergic neurons (MDNs), which are critical for the production and release of dopamine – a neurotransmitter essential for regulating movement, motivation, and learning. The loss of these vital neurons leads to the hallmark motor symptoms of PD, including tremors, bradykinesia (slowness of movement), rigidity, and postural instability, profoundly impacting patients’ quality of life and independence. Beyond motor symptoms, many individuals also experience non-motor symptoms such as cognitive impairment, sleep disturbances, and depression, adding to the complexity and burden of the disease.
Current treatments for Parkinson’s disease primarily focus on managing symptoms rather than halting or reversing the neurodegeneration. Levodopa, a dopamine precursor, remains the most effective symptomatic therapy, but its long-term use often leads to motor complications such as dyskinesia. Other approaches include dopamine agonists, MAO-B inhibitors, and surgical interventions like deep brain stimulation (DBS), which can offer significant relief for some patients but do not address the underlying cause of neuronal loss. The absence of a cure underscores the urgent global need for innovative therapeutic strategies, with regenerative medicine and cell therapy emerging as particularly promising avenues for potentially replacing the lost dopaminergic neurons and restoring function.
The Quest for High-Quality Neurons: Challenges in Regenerative Medicine
The concept of replacing damaged or lost neurons through cell transplantation has captivated neuroscientists for decades, offering a tantalizing prospect for treating PD and other neurodegenerative conditions. Early attempts at fetal tissue transplantation, while showing some promise, were fraught with ethical concerns, practical limitations in sourcing material, and inconsistent clinical outcomes, including the development of dyskinesias in some recipients. The advent of induced pluripotent stem cell (iPSC) technology, which allows adult cells to be reprogrammed into an embryonic-like state and subsequently differentiated into specific cell types like MDNs, revolutionized the field. iPSCs offer a patient-specific and ethically sound source of cells, paving the way for personalized regenerative therapies.
However, the journey from iPSC to therapeutically viable MDN graft is fraught with challenges. A critical hurdle lies in ensuring the purity and functional identity of the lab-grown neurons. For cell therapy to be safe and effective, the transplanted cells must not only be the correct cell type (e.g., MDNs for Parkinson’s) but also free from contamination by unwanted cell populations. The presence of "off-target" cells—cells that differentiate into unintended brain regions or non-neuronal cell types—poses significant risks. These risks include the potential for tumor formation (teratomas), the generation of inappropriate neural circuits that could worsen symptoms or cause new neurological issues, or simply a reduction in the efficacy of the graft due to a lower proportion of functional therapeutic cells. Therefore, robust methods for identifying, characterizing, and purifying specific neuronal populations are paramount for translating stem cell research into safe and efficacious clinical treatments.
BrainSTEM: A Novel Approach to Charting the Developing Brain
Addressing these critical challenges, the research team developed an innovative two-step mapping approach named BrainSTEM, an acronym for "Brain Single-cell Two tiEr Mapping." This sophisticated methodology leverages the power of single-cell genomics to provide an unparalleled view into neural development. Single-cell RNA sequencing (scRNA-seq), the underlying technology, allows researchers to analyze the gene expression profiles of individual cells, providing a "molecular fingerprint" that distinguishes one cell type from another and reveals its developmental trajectory. This is a significant advancement over bulk RNA sequencing, which averages gene expression across millions of cells, obscuring the unique characteristics of individual cell populations.
In a collaborative effort that included partners such as the University of Sydney, the BrainSTEM team embarked on an ambitious project to profile nearly 680,000 cells derived from the fetal human brain. The decision to map the fetal brain is strategic, as it represents the natural blueprint for how diverse neural cell types develop and interact, providing a crucial reference point for understanding disease mechanisms and for guiding the differentiation of stem cells into specific neuronal populations. The first tier of BrainSTEM involved a broad, comprehensive mapping of the entire cellular landscape of the fetal brain, creating a foundational atlas of neural diversity.
The second, higher-resolution tier of BrainSTEM then targeted the midbrain with added precision, specifically pinpointing the development of dopaminergic neurons. This focused approach allowed the researchers to create an exquisitely detailed "comprehensive reference map" of MDN development. This map is not merely an observational tool; it is designed to serve as a global standard, providing an objective benchmark for evaluating how accurately laboratory-generated midbrain models—derived from iPSCs or other stem cell sources—match the complex biology of real human MDNs. By comparing the genetic signatures and developmental trajectories of lab-grown cells against this gold-standard reference, researchers can now rigorously assess the purity, identity, and maturity of their cell products, a critical step towards clinical translation.
Unveiling Key Findings: Purity and Precision
The application of BrainSTEM yielded a crucial and immediately actionable finding: several methods currently used in laboratories worldwide to grow midbrain cells also inadvertently generated unwanted cells originating from other brain regions. These "off-target" populations, often subtle and difficult to detect with less precise methods, were unequivocally identified by BrainSTEM’s high-resolution analysis. This discovery carries profound implications for the development of safe and effective cell therapies.
The presence of such unwanted cells in preparations intended for transplantation can lead to a host of problems, ranging from reduced therapeutic efficacy to potentially harmful side effects, including the formation of tumors or the disruption of existing neural circuits. The study’s findings therefore serve as a critical alert to the scientific community, indicating that both experimental protocols for cell differentiation and the subsequent data analysis pipelines used to characterize these cells need significant refinement. BrainSTEM, by precisely distinguishing even subtle off-target cell populations, provides the necessary diagnostic power to identify these impurities, enabling researchers to optimize their differentiation protocols to achieve higher purity and specificity. This rigorous validation step is indispensable for increasing the safety and efficacy of future cell therapies and minimizing potential adverse outcomes for patients.
Expert Perspectives on the Breakthrough
The significance of the BrainSTEM atlas has been underscored by the research team members themselves, who envision its broad impact on neuroscience and regenerative medicine.
Dr. Hilary Toh, an MD-PhD candidate from the Neuroscience & Behavioural Disorders program at Duke-NUS Medical School and one of the first authors of the paper, emphasized the practical utility of the atlas: "Our data-driven blueprint helps scientists produce high-yield midbrain dopaminergic neurons that faithfully reflect human biology. Grafts of this quality are pivotal to increasing cell therapy efficacy and minimizing side effects, paving the way to offer alternative therapies to people living with Parkinson’s disease." Her statement highlights the direct clinical relevance of the work, focusing on the production of safer and more effective therapeutic cells.
Dr. John Ouyang, Principal Research Scientist from Duke-NUS’ Centre for Computational Biology and a senior author of the study, pointed to the future potential, particularly in the realm of artificial intelligence: "By mapping the brain at single-cell resolution, BrainSTEM gives us the precision to distinguish even subtle off-target cell populations. This rich cellular detail provides a critical foundation for AI-driven models that will transform how we group patients and design targeted therapies for neurodegenerative diseases." This perspective opens avenues for personalized medicine, where AI could leverage this detailed cellular information to predict disease progression and optimize treatment strategies for individual patients.
Assistant Professor Alfred Sun from Duke-NUS’ Neuroscience & Behavioural Disorders programme, also a senior author of the paper, underscored the transformative nature of BrainSTEM: "BrainSTEM marks a significant step forward in brain modeling. By delivering a rigorous, data-driven approach, it will speed the development of reliable cell therapies for Parkinson’s disease. We’re setting a new standard to ensure the next generation of Parkinson’s models truly reflects human biology." His remarks emphasize the establishment of a new benchmark for scientific rigor and the acceleration of therapeutic development.
Professor Patrick Tan, Senior Vice-Dean for Research at Duke-NUS, provided a broader institutional perspective on the achievement: "This study redefines the benchmark—establishing multi-tier mapping as essential for capturing cellular detail in complex biological systems. By revealing how the human midbrain develops in such detail, we will accelerate Parkinson’s research and cell therapy, delivering better care and offer hope to people living with the disease." Professor Tan’s comments reinforce the foundational nature of BrainSTEM, positioning it as a critical tool for advancing understanding across various complex biological systems, with immediate implications for Parkinson’s research and patient care.
Broader Implications and Future Directions
The BrainSTEM atlas and its accompanying methodology are poised to have far-reaching implications across multiple facets of neuroscience and biomedical research.
Standardization in Neuroscience Research: By providing a "comprehensive reference map" and a rigorous evaluation framework, BrainSTEM sets a new global standard for assessing the quality and fidelity of lab-grown neural cells. This standardization is crucial for ensuring reproducibility across different laboratories, facilitating more reliable comparisons of experimental results, and accelerating the translation of research findings into clinical applications. It means that researchers worldwide can now speak a common language when characterizing their stem cell-derived neurons, leading to more robust and trustworthy preclinical data.
Accelerating Drug Discovery and Preclinical Testing: Better, more biologically accurate human neuronal models mean more reliable platforms for drug screening. Pharmaceutical companies can use cells validated by BrainSTEM to test potential new therapies for Parkinson’s and other neurological conditions with greater confidence, potentially reducing the high attrition rates seen in neuroscience drug development. The ability to identify and remove off-target cell populations ensures that drug effects are measured on the intended therapeutic cells, leading to more precise and interpretable results.
Advancing Personalized Medicine with AI: Dr. Ouyang’s mention of AI-driven models highlights a future where personalized medicine for neurodegenerative diseases could become a reality. By integrating the detailed cellular and genetic information from BrainSTEM with patient-specific data, AI algorithms could help stratify patients, predict individual responses to therapies, and even design bespoke cell lines or drug regimens tailored to an individual’s unique genetic and cellular profile. This move towards precision medicine holds the promise of more effective treatments with fewer side effects.
Fostering Open Science and Global Collaboration: In a move that champions open science, the researchers plan to release their brain atlases as open-source references and provide the multi-tier mapping approach as an "out-of-the-box" package. This commitment to accessibility will empower labs worldwide to utilize BrainSTEM’s capabilities, democratizing access to cutting-edge tools. Because BrainSTEM is designed to be applicable to isolate any cell type in the brain, its utility extends far beyond Parkinson’s disease. Researchers studying Alzheimer’s disease, stroke, autism spectrum disorders, or other neurological conditions can adapt the methodology to deepen their insights, streamline their workflows, and accelerate discovery across the entire spectrum of neuroscience. This open-source approach fosters a collaborative research environment, facilitating faster progress through shared knowledge and tools.
Ethical Considerations and Clinical Translation: While BrainSTEM marks a significant scientific achievement, the path to clinical translation for cell therapies remains complex. The ethical imperative for robust safety and efficacy testing cannot be overstated. BrainSTEM provides a powerful tool for preclinical validation, ensuring that only the most rigorously characterized and purest cell products advance to human trials. This meticulous approach is vital to build public trust and ensure that novel therapies ultimately deliver on their promise without introducing unforeseen risks to patients.
Funding and Institutional Support
This groundbreaking work received substantial support from several key programs and philanthropic organizations. These included the USyd-NUS Ignition Grant, a testament to the strong international collaboration between the University of Sydney and Duke-NUS, and the Duke-NUS Parkinson’s Research Fund, made possible through a generous donation by The Ida C. Morris Falk Foundation. Such funding mechanisms are crucial for enabling ambitious, high-impact research projects that push the boundaries of medical science.
Duke-NUS Medical School continues to solidify its position as a leading institution in medical research and education, consistently demonstrating its commitment to improving patient care through scientific innovation. This study exemplifies the institution’s dedication to unraveling fundamental brain mechanisms and developing novel therapeutic strategies for neurological conditions, offering renewed hope to millions affected by these devastating diseases globally.