This unprecedented atlas identifies nearly every cell type within the intricate neural architecture, meticulously recording their unique genetic signatures and elucidating the complex ways these cells grow and interact. Beyond this foundational mapping, the research rigorously compares leading laboratory methods for producing high-quality neurons, a critical step forward in the quest to develop new, effective therapies for debilitating brain conditions such as Parkinson’s disease. The collaborative effort, spearheaded by Duke-NUS Medical School in Singapore and involving partner institutions including the University of Sydney, culminates in the innovative Brain Single-cell Two tiEr Mapping (BrainSTEM) approach, published in the esteemed journal Science Advances. This work is poised to redefine standards in neuroscience research and accelerate the translation of laboratory discoveries into clinical solutions.
The Urgent Need for Breakthroughs in Neurodegenerative Disease
Parkinson’s disease represents a formidable challenge to global health, standing as Singapore’s second most common neurodegenerative condition, affecting approximately three in every 1,000 individuals aged 50 and above. Globally, the prevalence is even more staggering, with estimates suggesting over 10 million people are living with the disease. The core pathology of Parkinson’s lies in the progressive degeneration of midbrain dopaminergic neurons, specialized nerve cells located in the substantia nigra region of the brain. These neurons are crucial for producing dopamine, a neurotransmitter vital for regulating movement, motivation, and learning. Their loss leads to the characteristic motor symptoms of Parkinson’s, including tremors, bradykinesia (slowness of movement), rigidity, and postural instability, significantly impairing a patient’s quality of life.
Current treatments, primarily levodopa, aim to manage symptoms by replenishing dopamine levels or mimicking its effects. While effective in the early stages, these therapies often become less efficacious over time, leading to motor fluctuations and dyskinesias—involuntary movements—as the disease progresses. Other interventions, such as deep brain stimulation (DBS), offer relief for some patients but are invasive surgical procedures and not universally suitable. The fundamental limitation of existing treatments is their inability to halt or reverse the neurodegeneration; they merely alleviate symptoms. This critical gap underscores the urgent need for restorative therapies, particularly those focused on replacing or regenerating the damaged dopaminergic neurons. Cell therapy, which involves transplanting lab-grown neurons into the brain, has emerged as a promising avenue, but its development has been hampered by significant challenges in generating cells that accurately mimic their human counterparts.
BrainSTEM: A New Paradigm for Brain Mapping
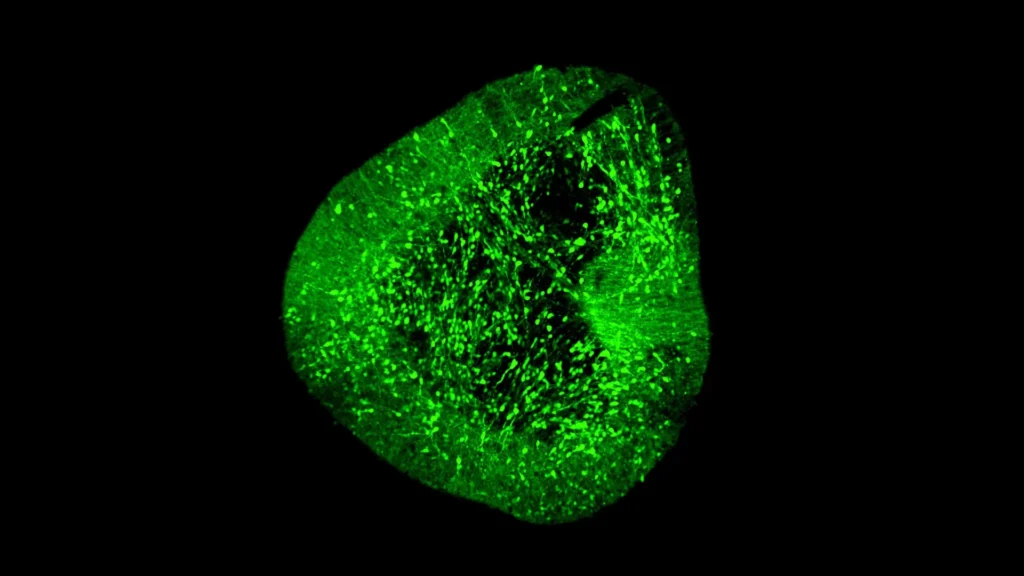
Addressing the complexities of neuronal development and the challenges of producing therapeutic cells, the Duke-NUS team, in collaboration with international partners, developed BrainSTEM. This innovative two-step mapping approach is designed to clarify precisely how dopaminergic neurons form and mature, both in vivo (within the living organism) and in vitro (in laboratory settings). The initial phase of BrainSTEM involved an expansive profiling effort, analyzing nearly 680,000 individual cells meticulously isolated from the fetal human brain. This massive dataset allowed researchers to construct a comprehensive cellular landscape, charting the full diversity and developmental trajectories of various brain cell types during critical stages of formation. This broad-strokes map provides an essential context, allowing scientists to understand the wider cellular environment in which specific neuron types develop.
The second, higher-resolution projection of BrainSTEM then zeroes in with unparalleled precision on the midbrain region, specifically targeting the dopaminergic neurons. This focused approach enabled the researchers to pinpoint the exact genetic signatures, developmental markers, and interactive pathways that define these critical neurons. The resulting "comprehensive reference map" is not merely a descriptive tool; it has been immediately recognized as a global standard. This benchmark is crucial for evaluating the accuracy and fidelity of midbrain models generated in laboratories, ensuring that researchers can reliably assess how closely their in vitro generated neurons match the real human biology they are intended to replace or repair.
Unmasking the Imperfections of Lab-Grown Neurons
One of the most significant and immediately actionable findings reported in the Science Advances study reveals a critical flaw in current laboratory methodologies. The research found that several widely used methods for growing midbrain cells, while successfully generating some desired neurons, also inadvertently produced "off-target" cells originating from other brain regions. These unwanted cell populations pose a substantial risk for future cell therapies. If transplanted into a patient, non-dopaminergic cells could lead to unintended side effects, reduced therapeutic efficacy, or even adverse neurological outcomes. The ability of BrainSTEM to detect even subtle off-target cell populations at single-cell resolution represents a monumental leap in quality control for regenerative medicine.
Dr. Hilary Toh, an MD-PhD candidate from the Neuroscience & Behavioural Disorders program at Duke-NUS Medical School and one of the paper’s first authors, underscored the transformative potential of these findings. "Our data-driven blueprint helps scientists produce high-yield midbrain dopaminergic neurons that faithfully reflect human biology," she explained. "Grafts of this quality are pivotal to increasing cell therapy efficacy and minimizing side effects, paving the way to offer alternative therapies to people living with Parkinson’s disease." This insight is critical, as it indicates that both experimental protocols for cell differentiation and the subsequent data analysis pipelines need substantial refining. The study provides a clear directive for the scientific community: develop more stringent methods to detect, isolate, and remove such unintended cellular contaminants, thereby ensuring the purity and safety of therapeutic cell products.
The Role of AI and the Future of Personalized Medicine
The precision afforded by BrainSTEM extends far beyond quality control for cell therapies. Dr. John Ouyang, Principal Research Scientist from Duke-NUS’ Centre for Computational Biology and a senior author of the study, highlighted its broader implications for artificial intelligence (AI) in neuroscience. "By mapping the brain at single-cell resolution, BrainSTEM gives us the precision to distinguish even subtle off-target cell populations," Dr. Ouyang stated. "This rich cellular detail provides a critical foundation for AI-driven models that will transform how we group patients and design targeted therapies for neurodegenerative diseases."
This synergy between high-resolution biological data and advanced computational power is poised to revolutionize personalized medicine. Currently, neurodegenerative diseases like Parkinson’s are often treated with a "one-size-fits-all" approach, despite growing evidence of significant patient heterogeneity in disease progression and response to treatment. BrainSTEM’s ability to delineate precise cellular and genetic signatures can feed into AI algorithms, enabling them to identify distinct disease subtypes or individual patient profiles. This could lead to the development of highly individualized therapeutic strategies, where treatments are tailored not just to the disease but to the unique biological characteristics of each patient, maximizing efficacy and minimizing adverse effects. This shift promises a future where drug discovery is more targeted, clinical trials are more efficient, and patient outcomes are significantly improved.
Accelerating Discovery and Setting New Standards
Assistant Professor Alfred Sun from Duke-NUS’ Neuroscience & Behavioural Disorders programme, also a senior author, emphasized the study’s impact on the pace of research. "BrainSTEM marks a significant step forward in brain modeling. By delivering a rigorous, data-driven approach, it will speed the development of reliable cell therapies for Parkinson’s disease," he commented. "We’re setting a new standard to ensure the next generation of Parkinson’s models truly reflects human biology." This establishment of a "new standard" is a profound declaration, signaling a paradigm shift in how research in this field will be conducted and evaluated. It implies a move towards more stringent validation processes and a greater emphasis on biological fidelity in in vitro models.
In a move designed to maximize its global impact and accelerate scientific discovery, the researchers have committed to releasing their comprehensive brain atlases as open-source references. Furthermore, the multi-tier mapping approach of BrainSTEM will be provided as an "out-of-the-box" package. This open-access philosophy is crucial for democratizing research tools and fostering collaborative science. Because BrainSTEM is inherently adaptable and can be applied to isolate and characterize virtually any cell type in the brain, laboratories worldwide can leverage this technology. This will allow researchers to deepen their understanding of other neurological conditions, streamline their workflows by having a standardized reference, and accelerate the pace of discovery across the entire spectrum of neuroscience. From studying the intricacies of memory formation to understanding the cellular basis of psychiatric disorders, BrainSTEM offers a versatile platform for unprecedented insight.
Broader Impact and Implications for Global Health
The implications of the BrainSTEM study extend far beyond Parkinson’s disease, promising a ripple effect across various fields of neuroscience and medical research. The ability to precisely map and understand the development of any brain cell type opens doors for:
- Alzheimer’s and Other Dementias: Characterizing the early cellular changes and interactions that lead to neurodegeneration in Alzheimer’s, frontotemporal dementia, or Lewy body dementia, offering new targets for early intervention.
- Developmental Brain Disorders: Gaining deeper insights into conditions like autism spectrum disorder, schizophrenia, and epilepsy by mapping the atypical development or connectivity of specific neuronal populations.
- Drug Discovery: Providing highly accurate human-specific cellular models for high-throughput drug screening, significantly improving the predictive power of preclinical drug candidates and reducing the high failure rate in clinical trials for neurological drugs.
- Gene Therapy: Identifying specific cell types that are most vulnerable or most amenable to gene editing or gene delivery strategies, thereby optimizing the precision and efficacy of gene therapies.
- Regenerative Medicine Beyond Parkinson’s: Developing cell replacement therapies for other conditions where specific cell types are lost, such as motor neuron disease or spinal cord injury.
Professor Patrick Tan, Senior Vice-Dean for Research at Duke-NUS, aptly summarized the monumental nature of this achievement: "This study redefines the benchmark—establishing multi-tier mapping as essential for capturing cellular detail in complex biological systems. By revealing how the human midbrain develops in such detail, we will accelerate Parkinson’s research and cell therapy, delivering better care and offer hope to people living with the disease." This statement encapsulates the multifaceted impact of BrainSTEM, from its foundational scientific contribution to its tangible promise for improving patient lives.
The funding for this groundbreaking work highlights the collaborative spirit and strategic investment necessary for such ambitious projects. Support was received from diverse programs, including the USyd-NUS Ignition Grant, which fosters international research partnerships, and the Duke-NUS Parkinson’s Research Fund, made possible through a generous donation by The Ida C. Morris Falk Foundation. Such financial backing underscores the recognition of the critical importance of this research in addressing a major global health challenge.
Duke-NUS Medical School continues to solidify its reputation as a global leader in medical research and education, demonstrating an unwavering commitment to improving patient care through scientific innovation. The BrainSTEM study is a testament to this mission, advancing ongoing efforts not only to understand the fundamental mechanisms governing brain development and function but also to translate this knowledge into tangible, effective therapeutic strategies for a wide spectrum of neurological conditions. This work provides a beacon of hope for millions affected by brain disorders, promising a future where precision, efficacy, and personalized care become the hallmarks of neurological treatment.











